|
8/14/2023 0 Comments Viscosity of air at 10 cViscosity is a measure of a fluid’s resistance to flow. Gas viscosity is only weakly dependent on pressure near atmospheric pressure. The viscosity of gases near room temperature are in the centiPoise range, so that is a commonly used unit. The viscosity of a gas can be thought of as a measure of its resistance to flow and is measured in the CGS unit Poise = dyne sec/cm2. The dynamic viscosity decreases with an increasing altitude of up to 11′000 m, then stays constant to 25′000 m and increases from an altitude of more than 25′000 m. With an increasing altitude, the density of the air decreases. Does air viscosity change with pressure?Īt high pressures the viscosity increases with increasing pressure.ĭoes viscosity of air change with altitude? The ratio of shear stress to shear rate is a constant, for a given temperature and pressure, and is defined as the viscosity or coefficient of viscosity. Newton’s law of viscosity defines the relationship between the shear stress and shear rate of a fluid subjected to a mechanical stress. 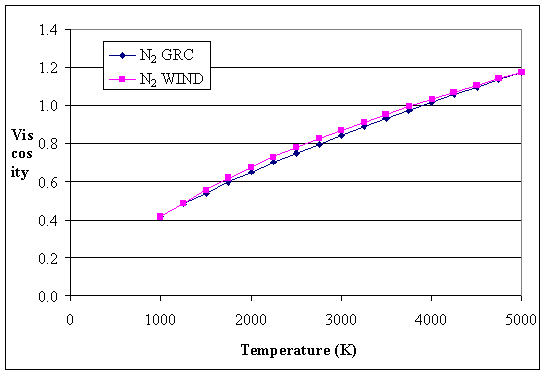
Thus, increasing the temperature of a gas will increase its momentum and thus the viscosity. This means that the viscosity of a gas is caused by the transfer of momentum from one gas molecule to another. In the case of a gas, viscosity results from jumping of molecules from one layer of a gas to another layer. As slower molecules collide with faster molecules, the slower molecules speed up and the faster molecules slow down. In gases, molecular collisions transfer momentum between fluid layers. The formula for density is d = m/v, where d is the density, m is the mass, and v is the volume. How do you find the properties of air?ĭensity is a measure of mass. In the category of fluids, liquids generally possess higher viscosities than gases. One concludes that, in general, solids have extremely high viscosities whereas fluids have low viscosities. Ice is 5 × 1016 times more viscous than air. Water, under normal temperatures, is about 50 times more viscous than air. Therefore, viscosity is dimensionally represented as. SI unit of Coefficient of viscosity (η) of liquid is kg m-1 s-1. What is the formula for viscosity of a liquid?Ĭoefficient of viscosity of a liquid is defined as the viscous force acting per unit area between two adjacent layers of a liquid with a velocity gradient normal to the direction of flow of the liquid. Molasses, for example, has a greater viscosity than water. The reciprocal of the viscosity is called the fluidity, a measure of the ease of flow. Viscosity, resistance of a fluid (liquid or gas) to a change in shape, or movement of neighbouring portions relative to one another. Hence, gaseous viscosity increases with temperature. Since the momentum transfer is caused by free motion of gas molecules between collisions, increasing thermal agitation of the molecules results in a larger viscosity. How does viscosity of air vary with temperature? The way to find the viscosity of a gas is to calculate the rate of z-direction (downward) transfer of x -momentum, as explained in the previous section but one. How do you measure the viscosity of a gas? 
Kinematic viscosity of air at 20☌ is given to be 1.6 × 10-5 m2/s. The kinematic viscosity of air at 15 ☌ is 1.48 × 10-5 m2 /s or 14.8 cSt. At 15 ☌, the viscosity of air is 1.81 × 10-5 kg/(m The viscosity of air depends mostly on the temperature. There are several formulas and equations to calculate viscosity, the most common of which is Viscosity = (2 x (ball density – liquid density) x g x a^2) ÷ (9 x v), where g = acceleration due to gravity = 9.8 m/s^2, a = radius of ball bearing, and v = velocity of ball bearing through liquid. 24 What is the viscosity of water? How do you calculate viscosity in chemistry?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
